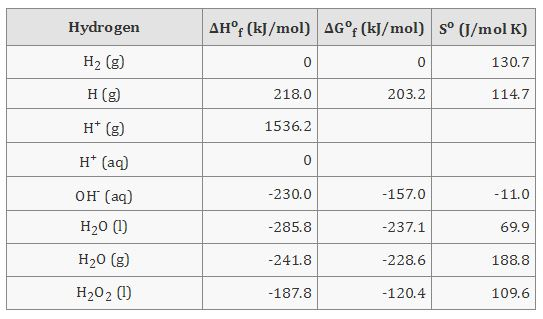
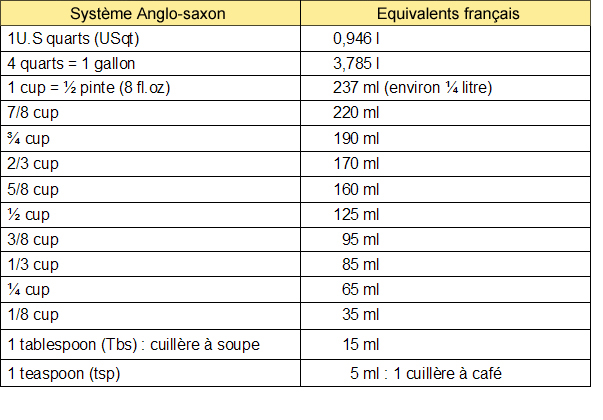
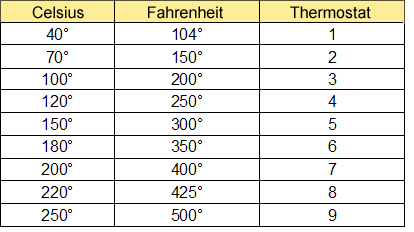
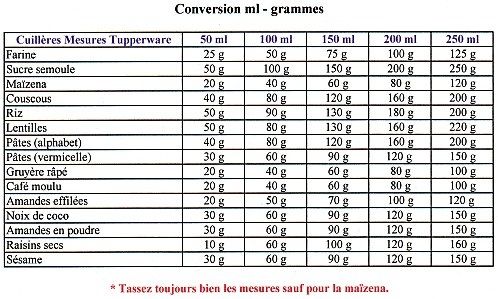
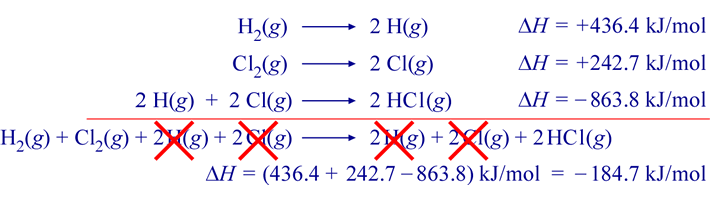0009905407 20 Torx Wheel Lug Nut Bolts 2 5/8" for Mercedes Benz CL E G GL GLA GLC GLE GLK ML R S Class - Wholesale Parts Express
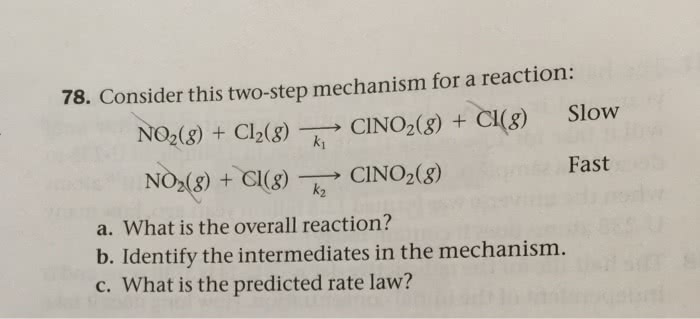
OneClass: 78. Consider this two-step mechanism for a reaction: Slow NO2(g) + Cl2(g) â†' CINO2(g) + Cl...
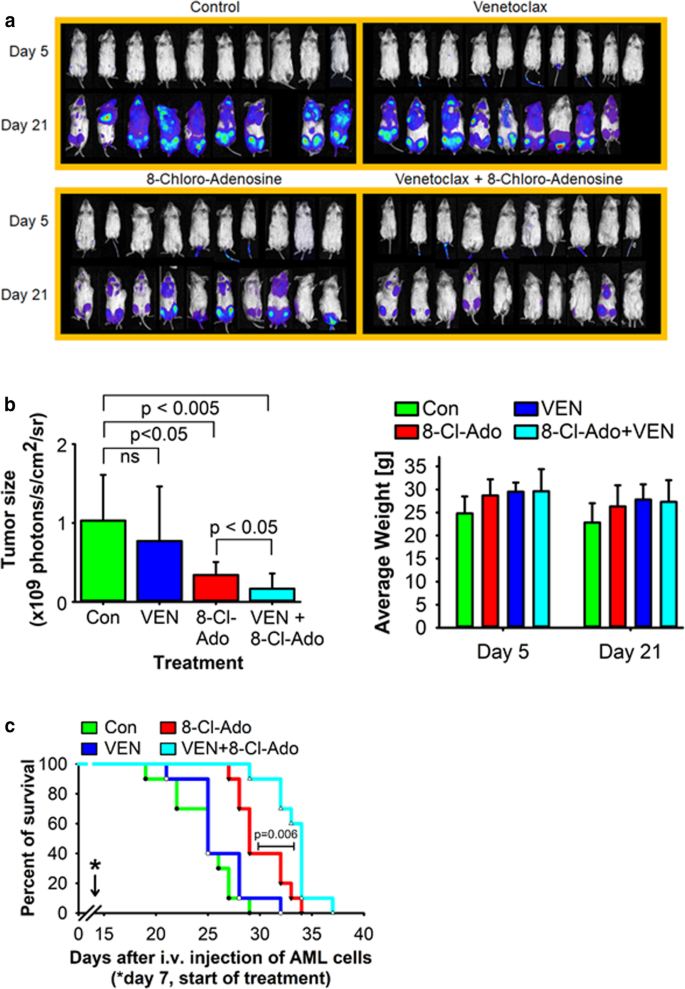
Targeting the metabolic vulnerability of acute myeloid leukemia blasts with a combination of venetoclax and 8-chloro-adenosine | Journal of Hematology & Oncology | Full Text

Chlorate(V) ions, ClO3^- , are produced in the redox reaction between chlorine and hot aqueoussodium hydroxide. Oxidation numbers can be used to help balance the equation for this reaction.What will be the

The reaction takes place in two steps as (i) NO(2) Cl ((g)) overset(K(1)) to NO(2(g)) +Cl((g)) (ii ) NO(2) Cl((g)) +Cl((g)) overset(K(2)) to NO(2(g)) +Cl(2(g)) Identify the reaction intermediate .