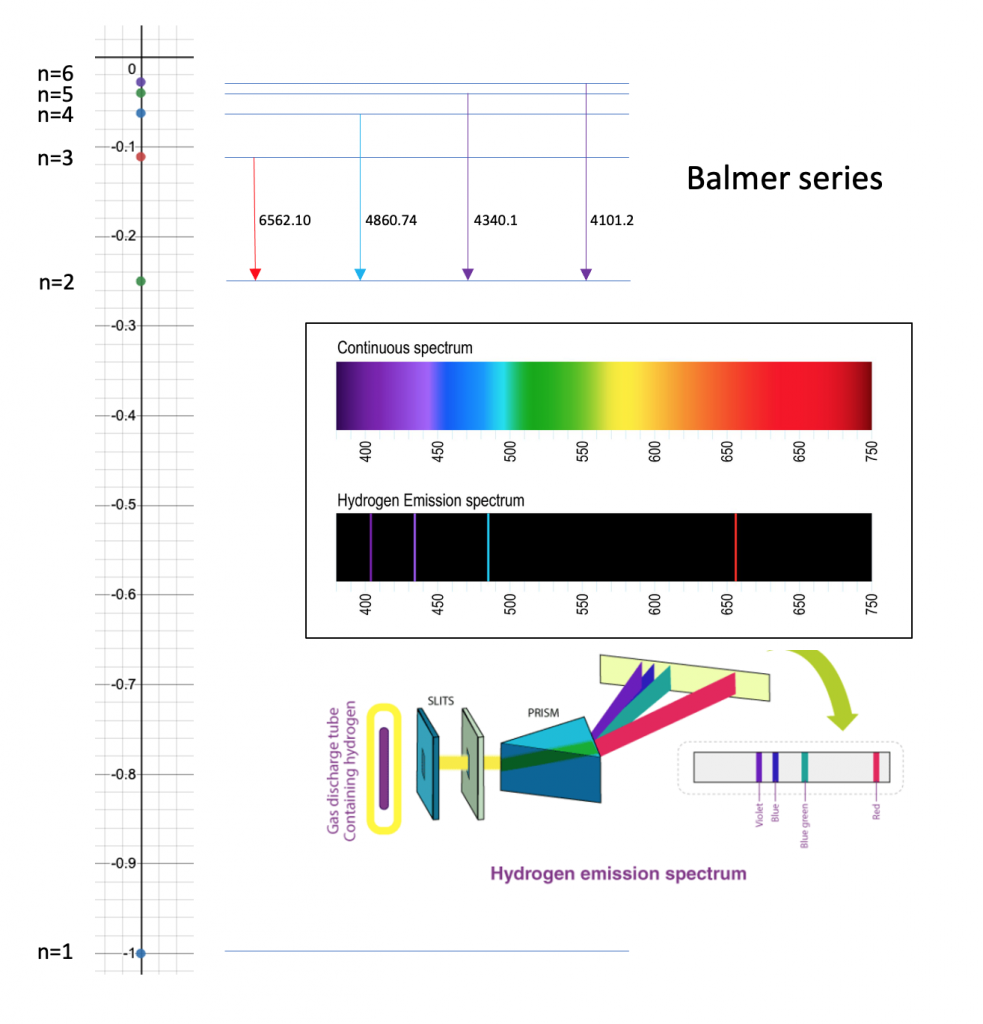
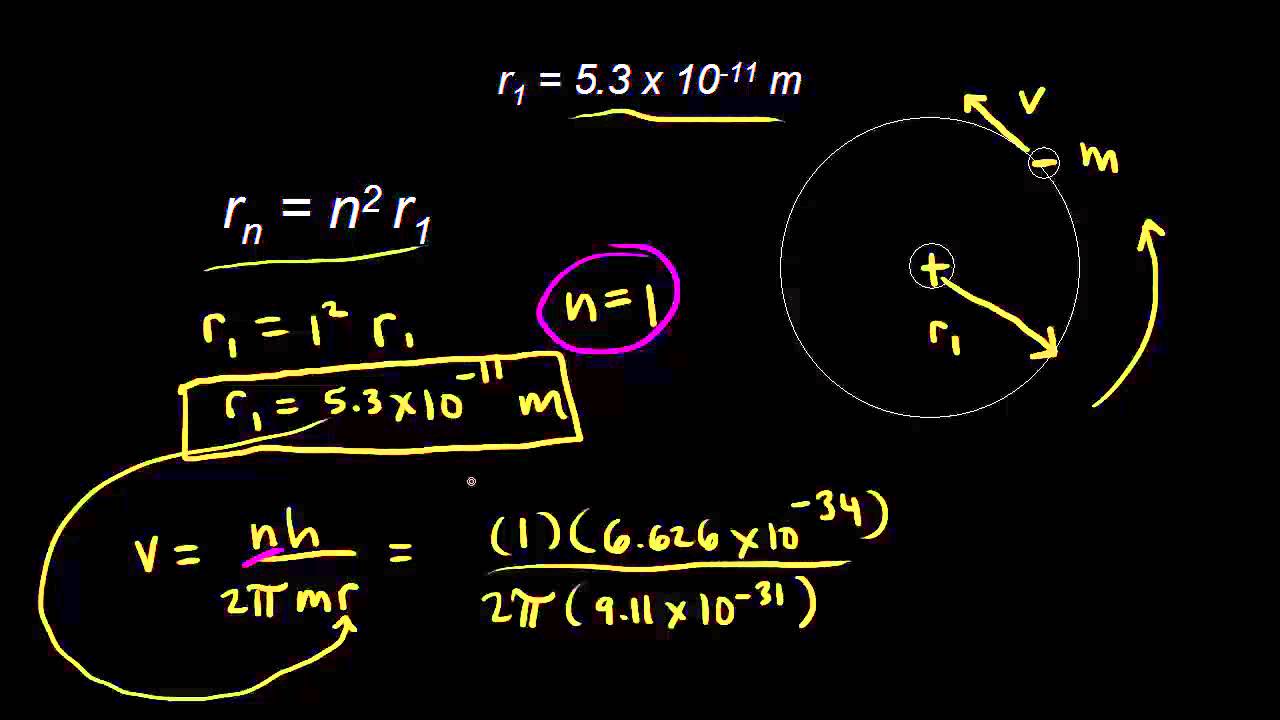The energy of an electron in the first Bohr orbit for hydrogen is - 13.6 eV. Which one of the following is a possible excited state for electron in Bohr orbit of

3 Calculate the wavelength in angstrom of thephoton that is emitted when an electron inBohr orbit n=2 - Brainly.in
According to Bohr's theory, the radius of the first orbit in a hydrogen atom is 0.528 Å. What is the radius of the fourth orbit? - Quora

6.25 | What is the radius, in angstroms, of the orbital of an electron with n = 8 in a hydrogen atom - YouTube

EDR(x; u) in compressed and expanded Kevan structures. Abscissa x is... | Download Scientific Diagram
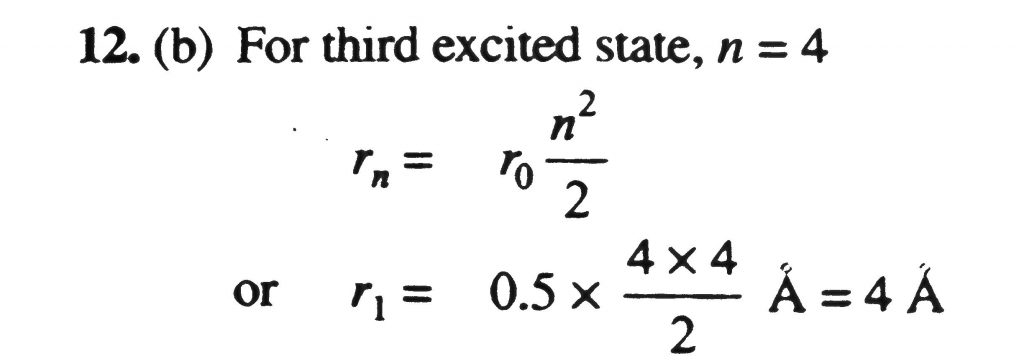
The radius of the Bohr orbit in the ground state of hydrogen atom is 0.5Å. The radius of the orbit of the electron in the third excited state of He+ will be -