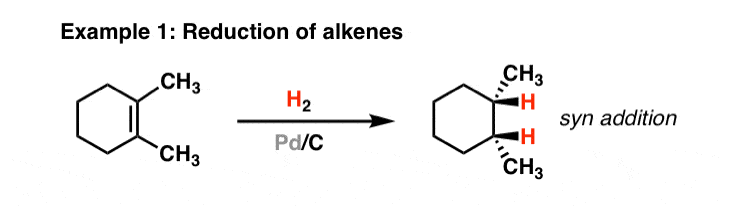
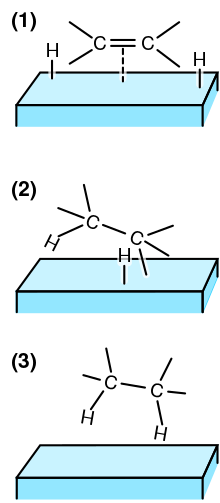
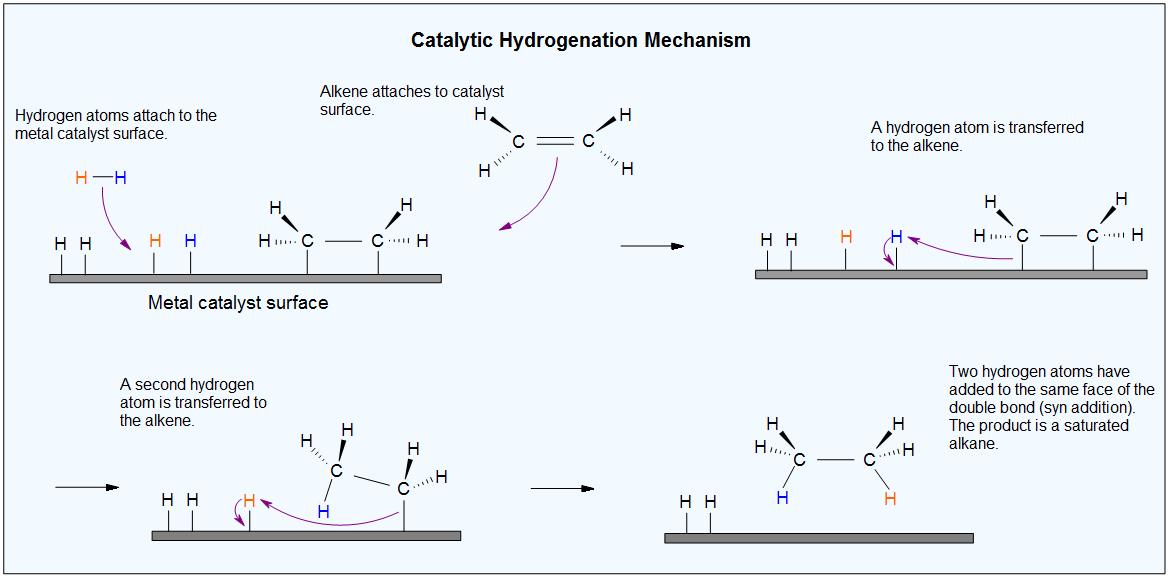
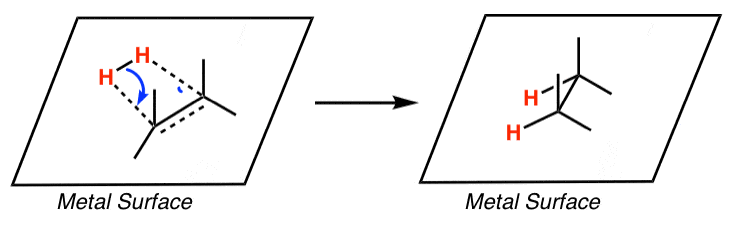
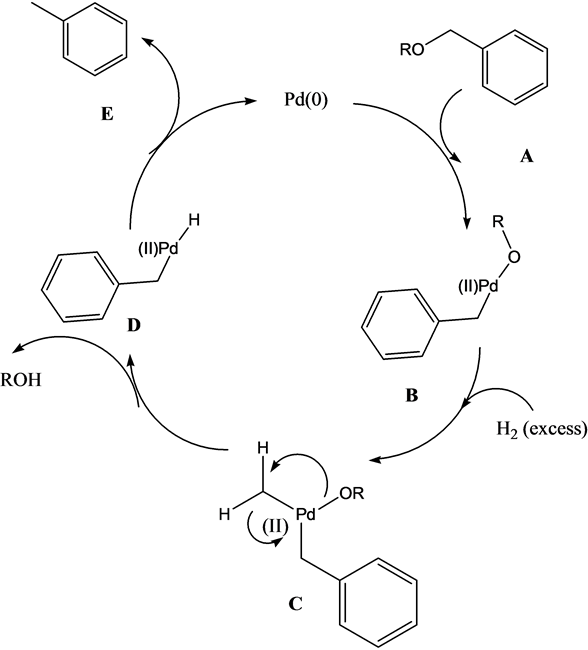Homogenous Pd-Catalyzed Asymmetric Hydrogenation of Unprotected Indoles: Scope and Mechanistic Studies | Journal of the American Chemical Society
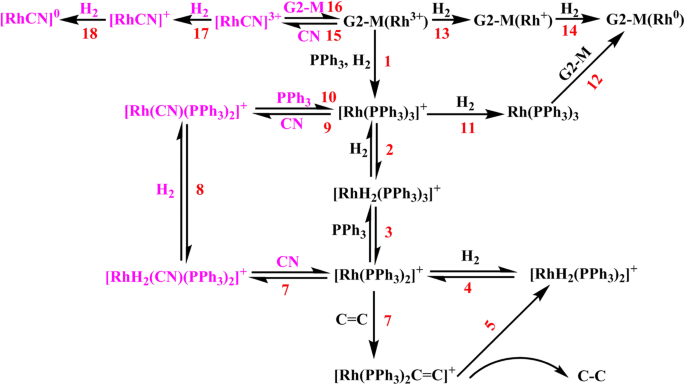
An insight into the catalytic hydrogenation mechanism of modified dendrimer-loaded rhodium ionic catalyst for unsaturated copolymer | SpringerLink

Mechanism of Pd(NHC)-Catalyzed Transfer Hydrogenation of Alkynes | Journal of the American Chemical Society
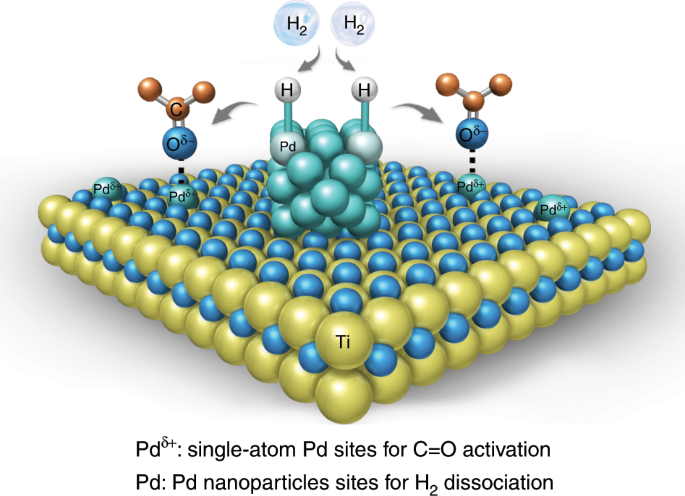
Titania supported synergistic palladium single atoms and nanoparticles for room temperature ketone and aldehydes hydrogenation | Nature Communications
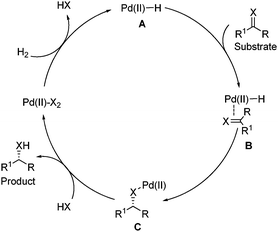
Homogeneous palladium -catalyzed asymmetric hydrogenation - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C2CS35333D

Facile Pd-catalyzed chemoselective transfer hydrogenation of olefins using formic acid in water - ScienceDirect

Figure 5 from Digital WPI Major Qualifying Projects ( All Years ) Major Qualifying Projects April 2018 Palladium On Carbon Catalyst | Semantic Scholar

Tetrahydroxydiboron-Mediated Palladium-Catalyzed Transfer Hydrogenation and Deuteriation of Alkenes and Alkynes Using Water as the Stoichiometric H or D Atom Donor
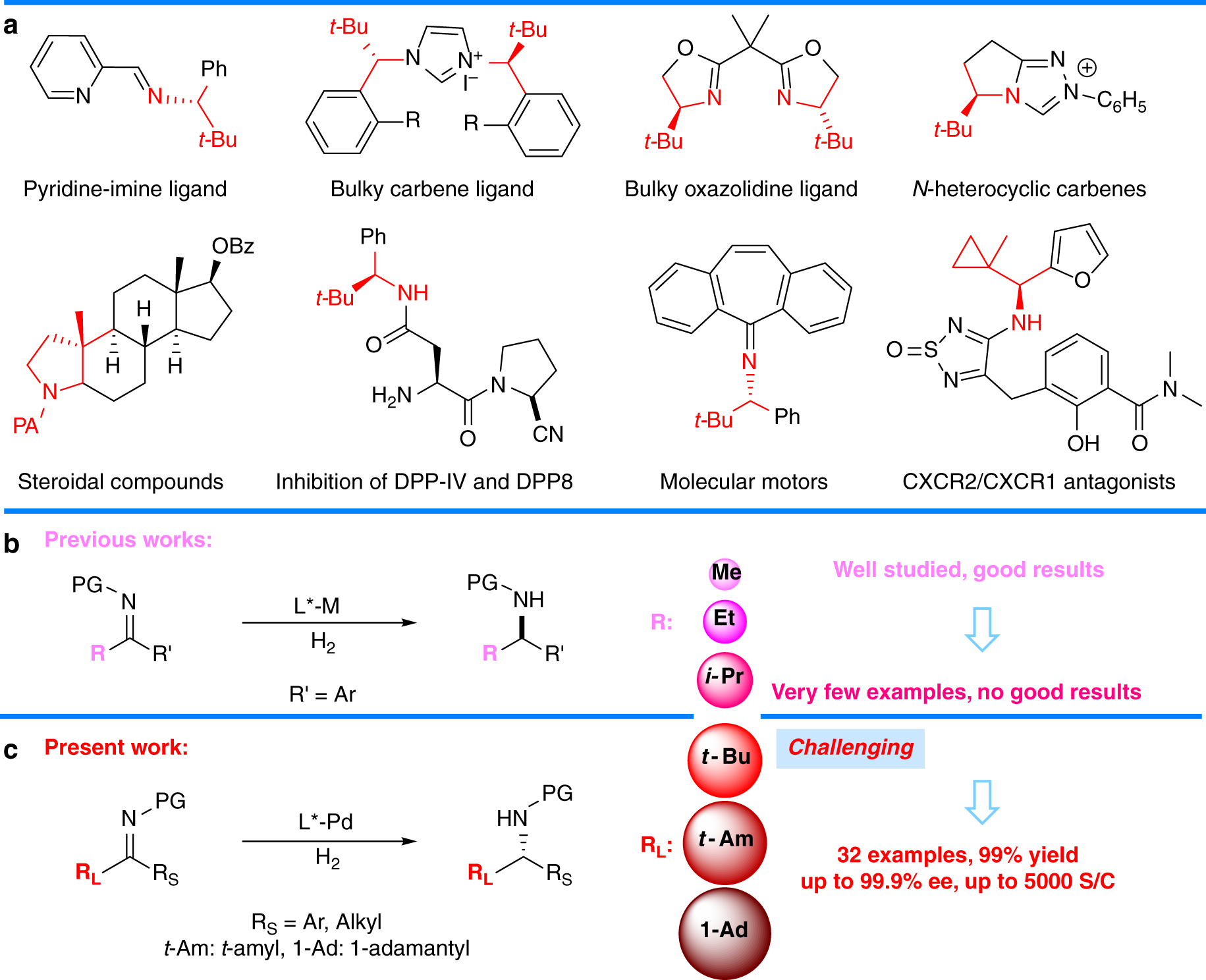
Pd(OAc)2-catalyzed asymmetric hydrogenation of sterically hindered N-tosylimines | Nature Communications
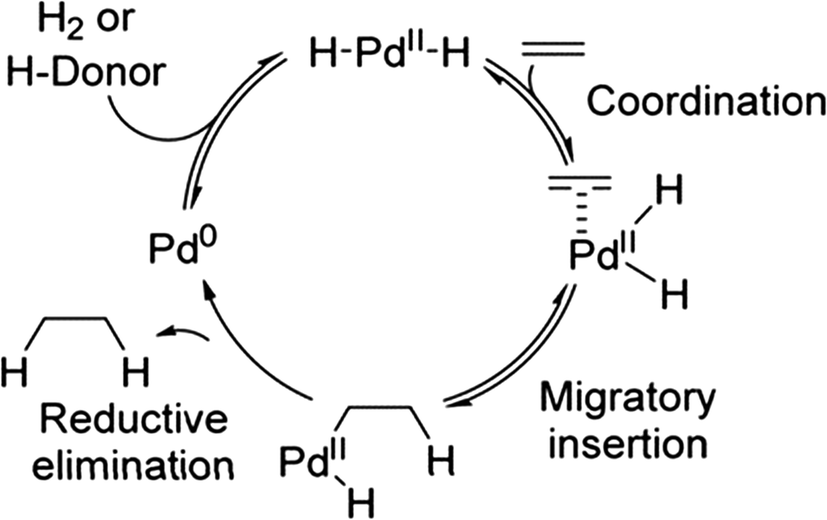
Towards high-performance heterogeneous palladium nanoparticle catalysts for sustainable liquid-phase reactions - Reaction Chemistry & Engineering (RSC Publishing) DOI:10.1039/D0RE00197J
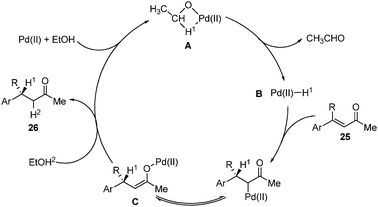
Homogeneous palladium -catalyzed asymmetric hydrogenation - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C2CS35333D
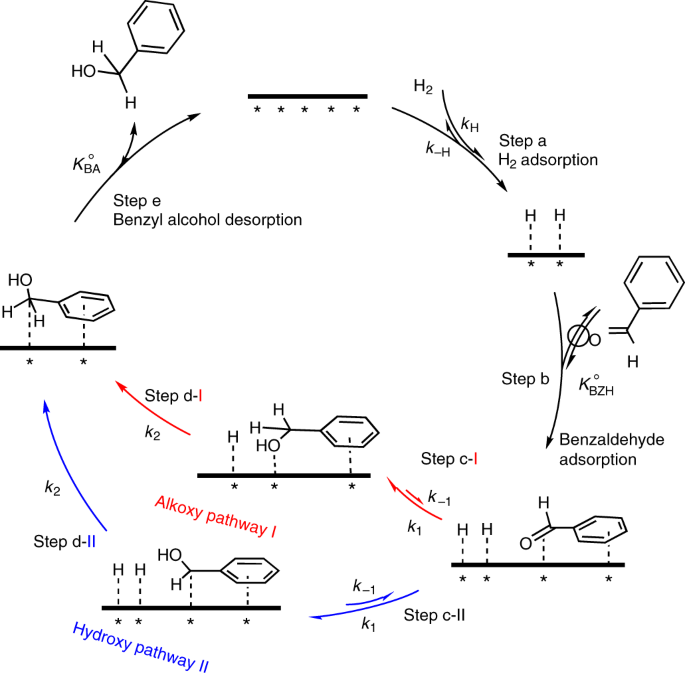
Critical role of solvent-modulated hydrogen-binding strength in the catalytic hydrogenation of benzaldehyde on palladium | Nature Catalysis
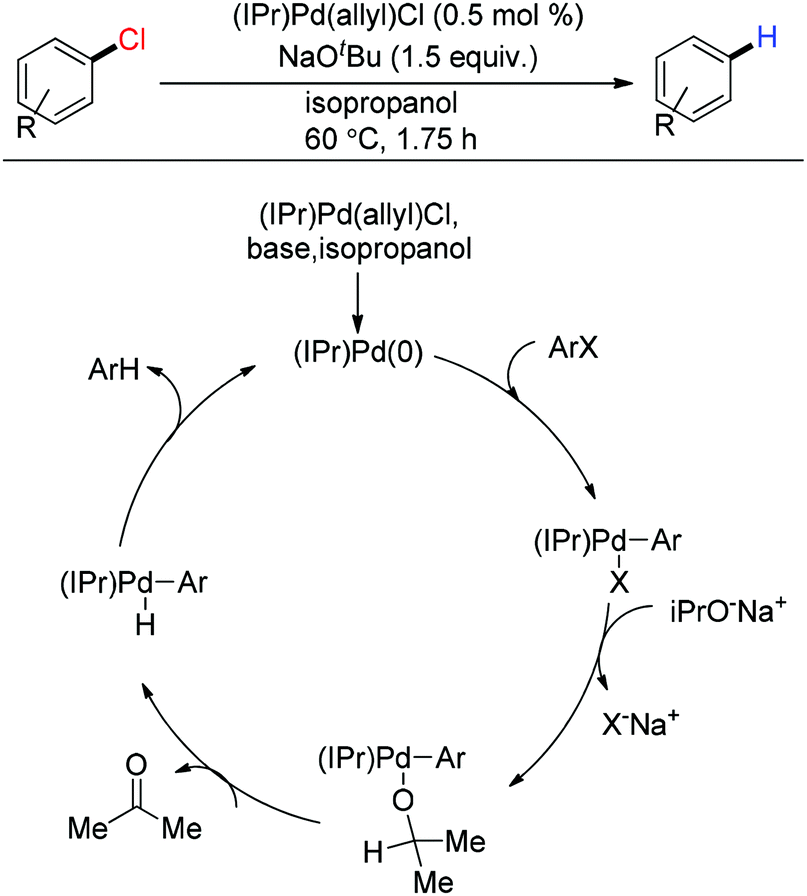
Metal catalyzed defunctionalization reactions - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB01949D

Homogeneous-like Alkyne Selective Hydrogenation Catalyzed by Cationic Nickel Confined in Zeolite | CCS Chem
![Palladium-catalyzed hydrogenation with use of ionic liquid bis(2-hydroxyethyl)ammonium formate [BHEA][HCO2] as a solvent and hydrogen source - ScienceDirect Palladium-catalyzed hydrogenation with use of ionic liquid bis(2-hydroxyethyl)ammonium formate [BHEA][HCO2] as a solvent and hydrogen source - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0040402013008594-fx1.jpg)