
Accelerated dinuclear palladium catalyst identification through unsupervised machine learning | Science

Benzyl Palladium Intermediates: Unique and Versatile Reactive Intermediates for Aromatic Functionalization - Zhang - 2021 - Advanced Synthesis & Catalysis - Wiley Online Library

Pd-Catalyzed Ring-Closing/Ring-Opening Cross Coupling Reactions: Enantioselective Diarylation of Unactivated Olefins | ACS Catalysis

Examples of five-membered ring palladium compounds 2.1-2.4 as catalysts... | Download Scientific Diagram
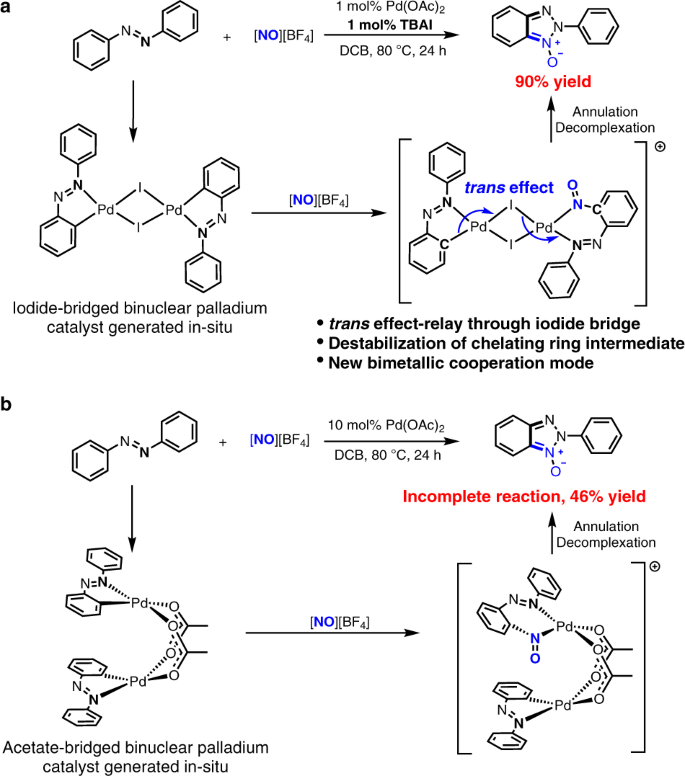
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry
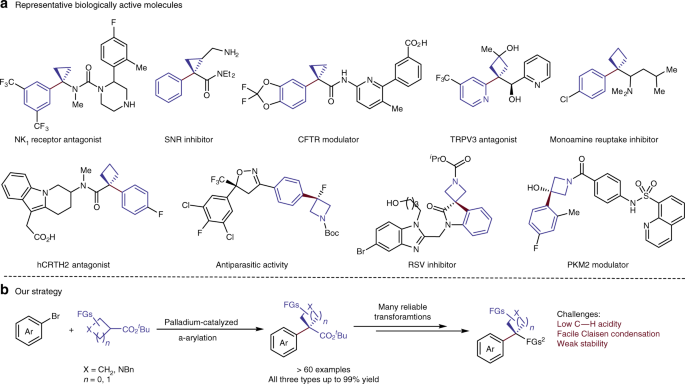
Palladium-catalyzed α-arylation for the addition of small rings to aromatic compounds | Nature Communications
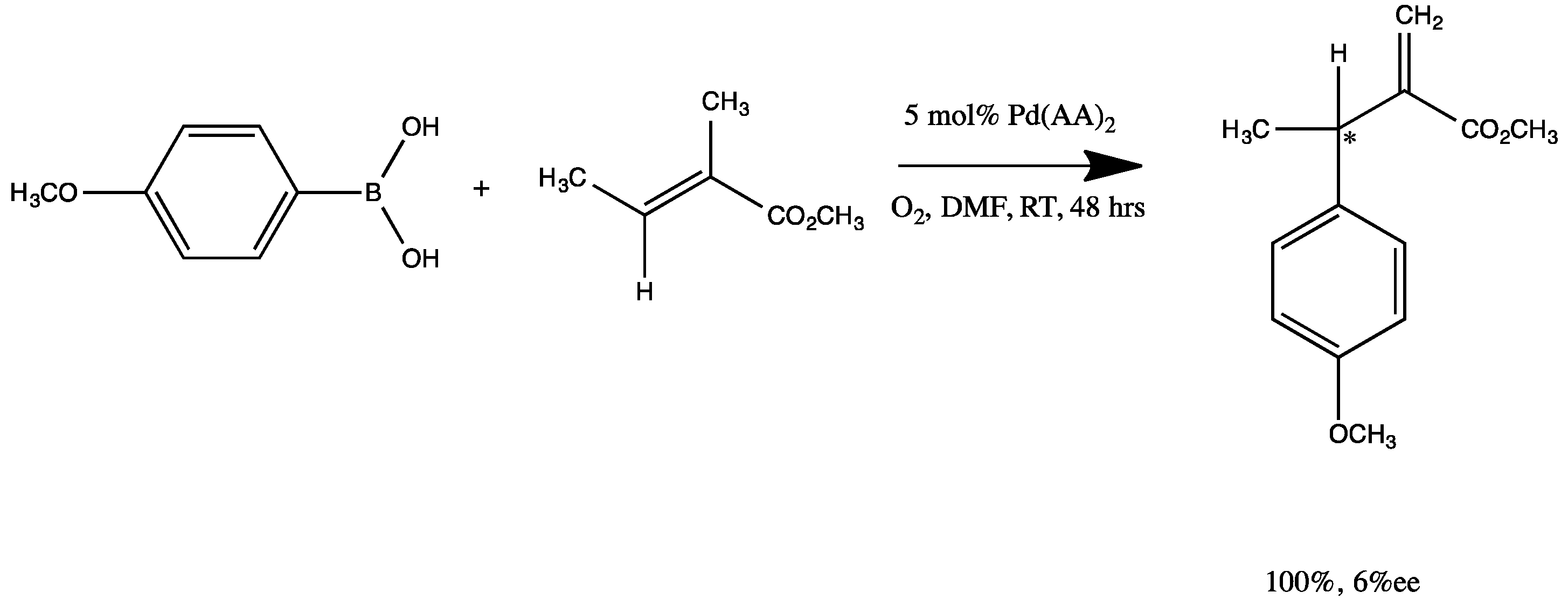
Catalysts | Free Full-Text | Synthesis, Structure, and Catalytic Reactivity of Pd(II) Complexes of Proline and Proline Homologs | HTML

Recoverable polystyrene-supported palladium catalyst for construction of all-carbon quaternary stereocenters via asymmetric 1,4-addition of arylboronic acids to cyclic enones - ScienceDirect

On the Mechanism of Palladium-Catalyzed Aromatic C−H Oxidation | Journal of the American Chemical Society

Palladium‐Catalyzed Ring‐Opening of 2‐Alkylidenecyclobutanols: Stereoselective Synthesis of γ,δ‐Unsaturated Ketones by C−C Bond Cleavage - Chen - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library

Palladium-Catalyzed Ring-Closing Reaction via C–N Bond Metathesis for Rapid Construction of Saturated N-Heterocycles | Journal of the American Chemical Society

Recoverable polystyrene-supported palladium catalyst for construction of all-carbon quaternary stereocenters via asymmetric 1,4-addition of arylboronic acids to cyclic enones - ScienceDirect

Examples of five-membered ring palladium compounds 2.1-2.4 as catalysts... | Download Scientific Diagram

Palladium(II)‐Catalyzed CH Activation/CC Cross‐Coupling Reactions: Versatility and Practicality - Chen - 2009 - Angewandte Chemie International Edition - Wiley Online Library

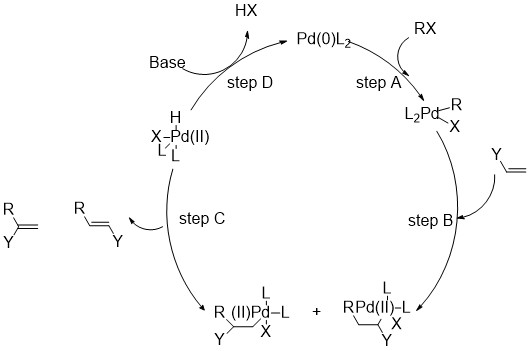




![Palladium Catalysts [Cross-coupling Reaction using Transition Metal Catalysts] | TCI AMERICA Palladium Catalysts [Cross-coupling Reaction using Transition Metal Catalysts] | TCI AMERICA](https://www.tcichemicals.com/assets/cms-images/03.gif)